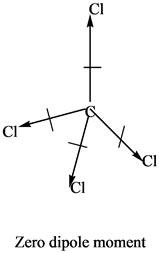
This result is in good agreement with the actual data: 2-methylpropane, boiling point = −11.7☌, and the dipole moment (μ) = 0.13 D methyl ethyl ether, boiling point = 7.4☌ and μ = 1. 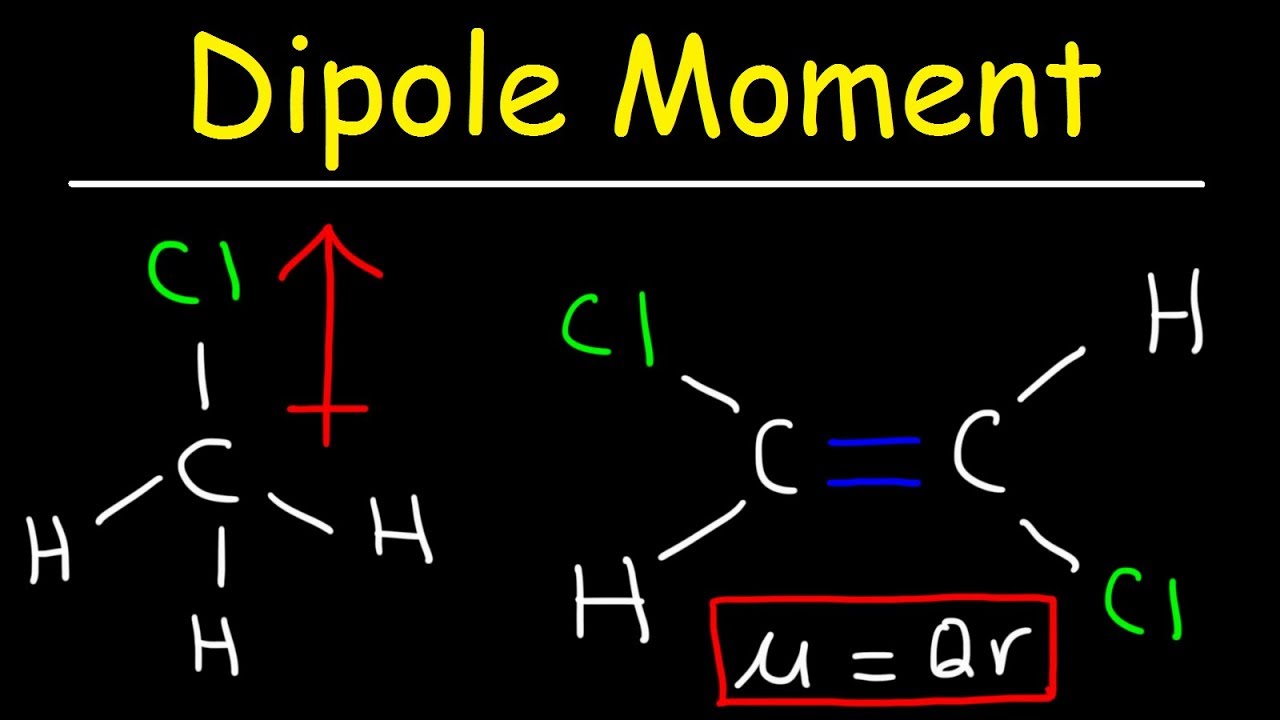
Thus we predict the following order of boiling points:Ģ-methylpropane < ethyl methyl ether < acetone. The C–O bond dipole therefore corresponds to the molecular dipole, which should result in both a rather large dipole moment and a high boiling point. A dipole moment is the product of the magnitude of the charge and the distance between the centers of the positive and negative charges. Thus a molecule such as H 2 O has a net dipole moment. In contrast, the H 2 O molecule is not linear (part (b) in Figure 9.8) it is bent in three-dimensional space, so the dipole moments do not cancel each other. As a result, the C–O bond dipoles partially reinforce one another and generate a significant dipole moment that should give a moderately high boiling point.Īcetone contains a polar C=O double bond oriented at about 120° to two methyl groups with nonpolar C–H bonds. As a result, the CO 2 molecule has no net dipole moment even though it has a substantial separation of charge. Transition dipole moment, the electrical. 
It should therefore have a very small (but nonzero) dipole moment and a very low boiling point.Įthyl methyl ether has a structure similar to H 2O it contains two polar C–O single bonds oriented at about a 109° angle to each other, in addition to relatively nonpolar C–H bonds. Dipole moment may refer to: Electric dipole moment, the measure of the electrical polarity of a system of charges. The first compound, 2-methylpropane, contains only C–H bonds, which are not very polar because C and H have similar electronegativities. The three compounds have essentially the same molar mass (58–60 g/mol), so we must look at differences in polarity to predict the strength of the intermolecular dipole–dipole interactions and thus the boiling points of the compounds. Dip ole moment is measured in Debye units, which is equal to the distance between the charges multiplied by the charge (1 Debye eq uals (3. If the dipole moments are of different magnitudes but in opposite directions, then the net dipole moment will be the vector sum (subtraction) of these dipole. Compounds with higher molar masses and that are polar will have the highest boiling points. Dipoledipole forces occur between molecules with permanent dipoles (i.e., polar molecules). When two electrical charges, of opposite sign and equal magnitude, are separated by a distance, a dipole is established.The size of a dipole is measured by its dipole moment ((mu)). Both molecules have a trigonal pyramidal shape however, their bond moments direction differs according to the atoms electronegativity since the net dipole. Their structures are as follows:Īsked for: order of increasing boiling pointsĬompare the molar masses and the polarities of the compounds. The attractive energy between two ions is proportional to 1/r, whereas the attractive energy between two dipoles is proportional to 1/r6.Īrrange ethyl methyl ether (CH 3OCH 2CH 3), 2-methylpropane, and acetone (CH 3COCH 3) in order of increasing boiling points. Similarly, on the axis of the dipole (but outside it), the field points in the same direction as the dipole, again getting weaker the further one gets from the charges.\): Relationships between the Dipole Moment and the Boiling Point for Organic Compounds of Similar Molar Mass Compound Notice that along the plane perpendicular to the axis of the dipole and midway between the charges, the direction of the electric field is opposite that of the dipole and gets weaker the further from the axis one goes. A dipole is a phenomena of charge displacement and dipole moment is its quantification. Ernest Zinck 9 years ago A moment is a measure of a turning force about an axis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
